Ulcerative Colitis
Ulcerative colitis is characterised by diffuse mucosal inflammation limited to colon. Disease extent can be broadly divided into distal and more extensive disease. “Distal" disease refers to colitis confined to the rectum(proctitis) orrectum and sigmoid colon (proctosimoiditis). More extensive disease includes “left sided colitis" (up to the splentic flexure). “Extensive colitis “(up to the hepatic flexure). andpancolitis (affecting the whole colon).
Exemptia journey- Data on Biosimilarity
Exemptia contains adalimumab which is biosimilar of originator’s adalimumab. It is an exact fingerprint match of originator’s adalimumab. Biosimilars are defined as ‘similar’ or ‘highly similar’ to the reference medicinal products (originator products) following the EMA (European Medicines Agency) and FDA (Food and Drug Administration, USA) regulatory guidelines. Biosimilars having the similar level of efficacy and safety compared to that of the originator products provide additional advantage to patients in terms of affordability and lost-cost of therapy, while expanding the patient access to therapies. Despite of its complex molecular nature, a target-directed approach for development has been proven highly successful to generate monoclonal antibodies similar to the originator products at the level of individual quality attributes with same functional properties.
Exemptia, termed as ZRC-3197, has been developed indigenously by Zydus-Cadila (Cadila Healthcare Ltd.), as biosimilar medicinal product of originator HUMIRA®. Originator HUMIRA®, a TNF blocker, is used for the treatment of rheumatoid arthritis, juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease and plaque psoriasis. Adalimumab, which serves the active substance of originator HUMIRA® is a fully human monoclonal IgG1 antibody produced recombinantly in genetically engineered Chinese Hamster Ovary (CHO) cells. Adalimumab is composed of 1330 amino acids with a molecular weight of about 148 kDa. Physicochemical and functional characterization of Exemptia and HUMIRA®, on a comparative basis to demonstrates biosimilarity between both.
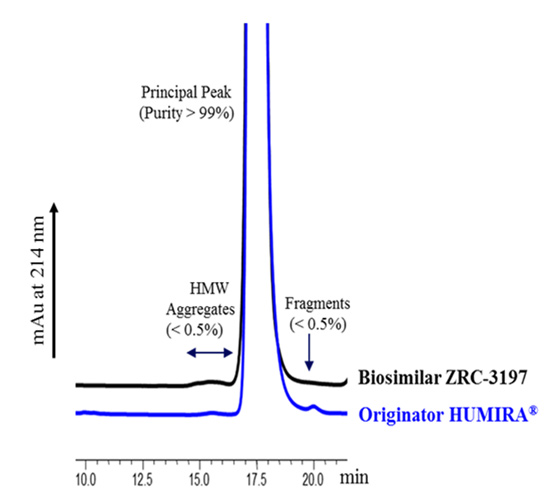
Comparison between the Exemptia and originator HUMIRA® by HP-SEC analysis
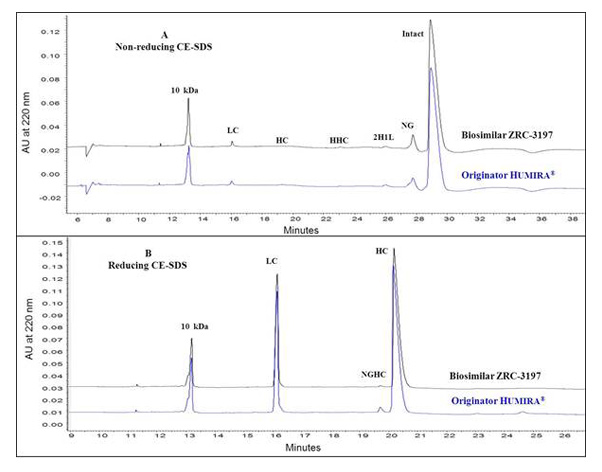
Comparative analysis of Exemptia and originator HUMIRA® for different size variants of adalimumab
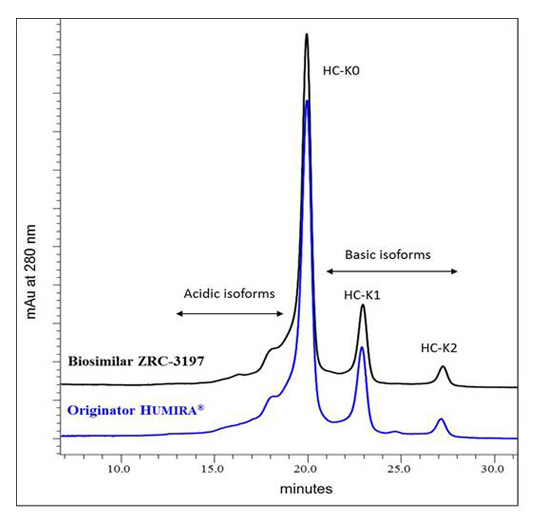
Charge heterogeneity profile comparison between the EXEMPTIA and HUMIRA
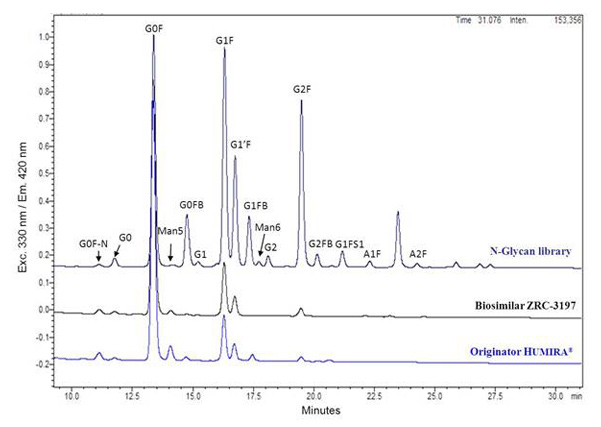
Glycans profile comparison between the EXEMPTIA and HUMIRA
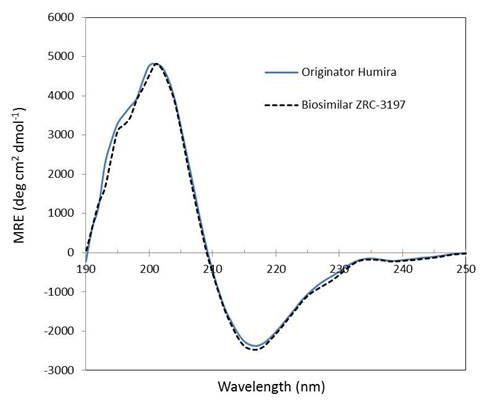
Secondary structure analysis
Pathophysiology of IBD
- According to the currently accepted hypothesis, ulcerative colitis and Crohn’s disease result from a dysregulated response of the mucosal immune system toward intraluminal antigens of bacterial origin in genetically predisposed persons. A number of pro-inflammatory cytokines, including interleukin-4 (IL-4), IL-5, IL-6, IL-10 and tumor necrosis factor-alpha (TNF-α), have been recognized to play a central role in mediating this immune response. Genetic and environmental factors induce impaired barrier function in the intestinal mucosa. Initiating triggers may involve infections in some patients. Altered barrier function subsequently induces the translocation of commensal bacteria and microbes.
- TNF-αlpha mediates multiple proinflammatory signals that play a central role in the pathogenesis of IBD, including neutrophil recruitment to local sites of inflammation, activation of both coagulation and fibrinolysis, and induction of granuloma formation. Increased numbers of TNF-αlpha producing cells are present in intestinal biopsy specimens from IBD patients.
- Moreover, enhanced secretion of TNF-αlpha from lamina propria mononuclear cells has been found in the intestinal mucosa of IBD patients. In Crohn’s disease tissues, TNF-αlpha positive cells have been found deeper in the lamina propria and in the submucosa, whereas TNF-αlpha immunoreactivity in ulcerative colitis is, mostly, located in subepithelial macrophages.
Use of Anti-TNFα Agents in the Treatment of Moderate to Severe Ulcerative Colitis
- Ulcerative colitis has an undulating pattern of disease activity, with 50% of all cases of UC estimated to be in remission at any moment in time. Although the inflammation of active UC is usually responsive to anti-inflammatory drugs to include: 5-ASA, corticosteroids, immunomodulators, in some cases it does not. In fact, 1/3rd of UC patients require colectomy in their lifetime, because of the development of cancer/dysplasia or colitisrefractory to medical therapy. Although surgery is considered a cure for UC, it is still associated with significant morbidity to include pouchitis (10% first year and up to 40% lifetime), impotence in males (10%), decreased fecundity in females (20-37%).
- Corticosteroids are effective in 50% of UC patients, while fewer than 20% will have no response to therapy. Responders to corticosteroids can be expected to remain in long term remission 50% of the time. Of those that relapse approximately 40-60% cases are responsive to immunomodulator therapy with azathioprine (AZA) or 6-mercaptopurine (6- MP).This leaves a substantial number of UC patients with medically refractory disease, and in need of a surgical solution or more effective medical treatment. Increased TNF from mucosal macrophages isolated from IBD lesions and increased TNF concentrations found in blood, mucosal tissue and stool of patients with UC has led to the consideration of Anti-TNF-α in the treatment of UC. Adalimumab is a fully human anti-TNF-α monoclonal antibody and consists of human derived heavy- and light-chain variable regions and human IgG1 constant region. Adalimumab was licensed for the treatment of moderately to severely active ulcerative colitis in adult patients who have had an inadequate response to conventional therapy including corticosteroids and 6-mercaptopurine or azathioprine, or who are intolerant to or have medical contraindications for such therapies.
Mechanism of Action
- The initial hypotheses leading to the development of anti-TNF-alpha antibodies as an IBD therapeutic tool resulted in the generation of data indicating that TNF-alpha represents a key cytokine involved in CD and UC. TNF-alpha is a transmembrane homotrimeric protein that is mainly expressed on, and secreted by, activated macrophages and T-lymphocytes. After proteolytic cleavage of the extracellular domain of the membrane-bound TNFalpha, these cells release soluble TNF-alpha. Both forms of TNF-alpha are able to bind to TNF-alpha receptor (TNFR)-1 or -2. After binding to TNFR-1 or -2, TNF-alpha activates several intracellular transduction pathways that lead to its physiological and pathological effects. TNF-alpha has been demonstrated to be an essential mediator in numerous immunological responses that include the downstream induction of ILs (eg, IL-1, IL-6, interferon-γ), adhesion molecules (eg, intracellular adhesion molecule-1), production of other inflammatory mediators, cell survival, differentiation, apoptosis, or necrosis. In inflammatory bowel disease (IBD), increased amounts of soluble and membranebound tumour necrosis factor (TNF) are produced by various immune and stromal cell populations, such as macrophages, dendritic cells (DCs), effector T cells, adipocytes etc.
- Adalimumab binds specifically to TNF-alpha and blocks its interaction with the p55 and p75 cell surface TNF receptors. Adalimumab also lyses surface TNF-expressing cells in vitro in the presence of complement. Adalimumab does not bind or inactivate lymphotoxin (TNF-beta). TNF is a naturally-occurring cytokine that is involved in normal inflammatory and immune responses.
- Adalimumab also modulates biological responses that are induced or regulated by TNF, including changes in the levels of adhesion molecules responsible for leukocyte migration [ELAM-1, VCAM-1, and ICAM-1]
Dosage & Administration
Adults with Crohn’s Disease or Ulcerative Colitis:
- The recommended induction dose is 160 mg at Week 0 (dose can be administered as four injections in one day or as two injections per day for two consecutive days), followed by 80 mg at Week 2.
- The recommended maintenance dose regimen is 40 mg every other week beginning at Week 4.
Efficacy
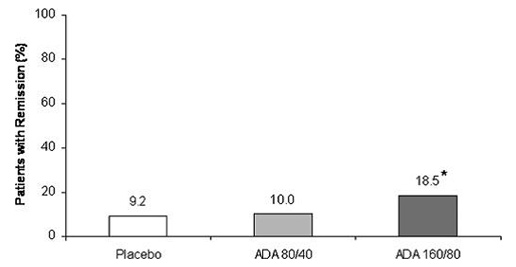
ULTRA 1 evaluated induction of clinical remission(Mayo score ≤2 with no subscore >1)
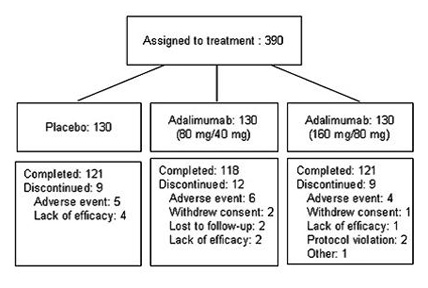
- Duration: 8-week, multicenter, randomized, double-blind, placebo-controlled study.
- Patients: 390 anti-TNF–naïve adult patients with moderate to severe UC defined as a Mayo score of ≥6 points and an endoscopy subscore of 2 or 3 despite concurrent or prior treatment with oral corticosteroids and/or azathioprine/6-mercaptopurine (AZA/6-MP)
- Concomitant Therapies: Patients in all study arms may have received stable doses of concomitant therapies that included AZA/6-MP, aminosalicylates, and oral corticosteroids.
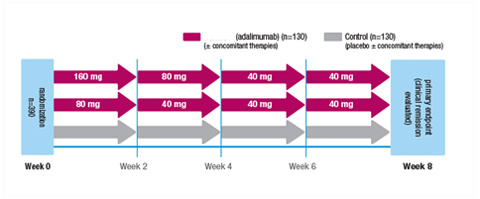
- Dosing:
- ADALIMUMAB 160 mg at Week 0, 80 mg at Week 2, and 40 mg at Weeks 4 and 6
(± concomitant therapies) Control (placebo ± concomitant therapies)
- Amendment 3 (A3): The original protocol was amended to include a second ADALIMUMAB induction group (ADALIMUMAB 80 mg at Week 0, 40 mg at Weeks 2, 4, and 6 ± concomitant therapies)
- Primary Endpoints: Percentage of patients achieving clinical remission (Mayo score ≤2 with no individual subscore >1) at Week 8 for each ADALIMUMAB arm.
- Analysis:
- Intent-to-treat (ITT-A3) study population using nonresponder imputation. ITT-A3 includes all randomized patients with confirmed UC enrolled under A3 or later who received ≥1 dose of study drug or control
- Patients with missing or incomplete data were classified as not in clinical remission
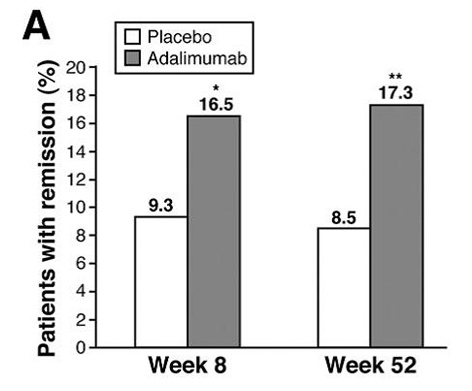
ULTRA 2 evaluated induction of and sustained clinical remission
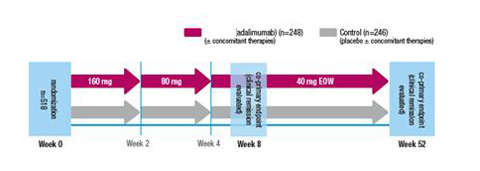
- Duration: 52-week, multicenter, randomized, double-blind, placebo-controlled study.
- Patients: 518 adult patients with moderate to severe UC for at least 3 months defined as a Mayo score of 6-12 points and an endoscopy subscore of at least 2 despite concurrent or prior therapy with oral corticosteroids and/or AZA/6-MP. Patients who lost response to or were intolerant to anti-TNF agents† were allowed entry into trial.
- Concomitant Therapies:
- Patients in both study arms may have received stable doses of concomitant therapies that included AZA/6-MP, aminosalicylates, and oral orticosteroids
- Corticosteroid dose could be tapered after Week 8
- Dosing: Patients were stratified by prior exposure to anti-TNF agents† and assigned to 1 of 2 treatment groups:
ADALIMUMAB 160 mg at Week 0, 80 mg at Week 2, and 40 mg every other week (EOW) starting at Week 4 (± concomitant therapies) Control (placebo ± concomitant therapies)
- Co-Primary Endpoints: Percentage of patients achieving clinical remission (Mayo score ≤2 with no individual subscore >1) at Week 8 and at Week 52
- Other Endpoints: Percentage of patients achieving sustained clinical remission (clinical remission at both Week 8 and Week 52)
- Analysis:
- Intent-to-treat (ITT) study population using nonresponder imputation. ITT includes all patients with confirmed UC who received 1 dose of study drug or control
- Patients with missing or incomplete data, or discontinued double-blind treatment, were classified as not in clinical remission
Outcomes
ULTRA 1-Approximately twice as many patients in the ADA 160/80 group achieved clinical remission at week 8 (primary endpoint) compared with patients in the placebo group (p=0.031; figure)
ULTRA 2(A)Proportion of patients with clinical remission at week 8 and week 52
Safety
IMPORTANT SAFETY INFORMATION for EXEMPTIA (adalimumab)1
SPECIAL WARNINGS AND PRECAUTIONS FOR USE
Infections
- Patients taking TNF-antagonists or with impaired lung function are susceptible to serious infections and therefore should be monitored throughout Adalimumab treatment including before, during and after treatment.
- Patients should only be treated with Adalimumab after active infections including chronic or localized infections are controlled. Risks and benefits of Adalimumab therapy should be reviewed prior to initiating the treatment in patients who have been exposed to tuberculosis (including travelled through high risk tuberculosis areas) or endemic mycoses such as histoplasmosis, blastomycosis and coccidioidomycosis.
- When patients develop a new infection while being treated with Adalimumab, a complete diagnostic evaluation should be performed and the patient should be closely monitored. Adalimumab should be discontinued in cases where a new serious infection or sepsis develops, and appropriate antifungal or antimicrobial therapy should be initiated to control the infection. Physicians should prescribe Adalimumab with caution after examining patient’s history to infections including the use of concomitant immunosuppressive medications.
Serious infections
- Patients receiving Adalimumab have encountered hospitalization, fatal outcomes, serious infections such as pneumonia, pyelonephritis, septic arthritis and septicemia due to bacterial, mycobacterial, invasive fungal, parasitic, viral, or other opportunistic infections such as listeriosis, legionellosis and pneumocystis.
Tuberculosis
- All patients must be examined in detail (including medical history) for both, active or inactive (latent) tuberculosis infection and current immunosuppressive therapy prior to administering Adalimumab as tuberculosis has been reported in patients receiving Adalimumab. The patient’s file should have records of tuberculin skin test and chest X-ray. Adalimumab therapy should not be initiated in those patients who have been diagnosed with active tuberculosis.
- An expert in tuberculosis should be consulted if latent tuberculosis is suspected when Adalimumab treatment is being considered.
- If latent tuberculosis is diagnosed, appropriate treatment must be started with anti-tuberculosis prophylaxis treatment before the initiation of Adalimumab, and in accordance with local recommendations. Use of anti-tuberculosis prophylaxis treatment should also be considered before the initiation of Adalimumab in patients with several or significant risk factors for tuberculosis despite a negative test for tuberculosis and in patients with a past history of latent or active tuberculosis in whom an adequate course of treatment cannot be confirmed.
- Patients should be instructed to seek medical advice for symptoms such as persistent cough, wasting/weight loss, low grade fever etc. that occur during or after Adalimumab treatment.
Other opportunistic infections
- Adalimumab treatment can result in opportunistic infections including invasive fungal infections in patients which if left untreated may prove fatal.
- Adalimumab treatment should be discontinued in patients who show symptoms of fever, malaise, weight loss, sweats, cough, dyspnea, and/or pulmonary infiltrates or other serious systemic illness with or without concomitant shock, and an invasive fungal infection should be suspected. Antifungal therapy should be initiated under extreme care and expertise in patients who show invasive fungal infections.
Hepatitis B reactivation
- Patients who are chronic carriers of Hepatitis B virus (i.e., surface antigen positive) can show reactivation of hepatitis B while being treated with TNF-antagonists such as Adalimumab, which in the past have sometimes resulted in fatal outcomes. Therefore, HBV infection test should be performed prior to administration of Adalimumab and an expert in treatment of hepatitis B should be consulted for patients who test positive for hepatitis B virus infection.
- Signs and symptoms for active HBV infection should be monitored in carriers of HBV throughout Adalimumab treatment and for several months after termination of therapy. Adalimumab treatment should be discontinued and anti-viral therapy should be initiated if HBV reactivation occurs in a patient.
Neurological events
- Rarely, Adalimumab and other TNF-antagonists have been shown to be associated with exacerbation or new onset of central or peripheral nervous system demyelinating disorders such as multiple sclerosis and Guillain-Barré syndrome. Thus, Adalimumab should be prescribed with caution in such cases.
Allergic reactions
- Only rare incidents of serious/non-serious allergic reactions such as anaphylaxis have been reported with Adalimumab treatment. Adalimumab treatment should be discontinued if serious allergic reactions such as anaphylaxis, allergic rash, fixed drug reaction, non-specified drug reaction or urticaria are observed in patients.
Immunosuppression
- Adalimumab treatment is not reported to be associated with depression of delayed-type hypersensitivity, depression of immunoglobulin levels, or change in enumeration of effector T-, B, - NK-cells, monocyte/macrophages, and neutrophils in clinical studies.
Malignancies
- Rare instances of malignancies such as lymphoma and leukemia have been seen in patients treated with TNF-antagonists. RA patients with long-standing highly active, inflammatory disease are at a higher risk of experiencing these malignancies.
- Malignancies in Adults: Cases of malignancies other than lymphoma that have been observed in patients with RA, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease and plaque psoriasis are breast cancer, colon cancer, prostate cancer, lung cancer and melanoma. Malignancies in the lung or head and neck were reported in patients treated with infliximab who had moderate to severe chronic obstructive pulmonary disease (COPD) and were heavy smokers. Therefore, caution should be exercised when using any TNF-antagonist in COPD patients, as well as in patients with increased risk for malignancy due to heavy smoking.
- Non-melanoma skin cancer: Prior to and during treatment with Adalimumab, patients should be examined for non-melanoma skin cancer especially if they have a history of extensive immunosuppressant therapy or psoriasis patients with a history of PUVA treatment.
- Lymphoma and Leukemia: The risk of lymphoma development is higher in patients with RA, other chronic inflammatory diseases particularly with highly active disease or chronic exposure to immunosuppressant therapies than general population even when a TNF blocker is not used. Acute and chronic leukemia were also reported with TNF-antagonist in RA and other indications.
- Malignancies, some fatal, have been reported among children, adolescents and young adults (up to 22 years of age) treated with TNF-antagonists (initiation of therapy ≤ 18 years of age), including Adalimumab. Approximately half the cases were lymphomas. The other cases represented a variety of different malignancies and included rare malignancies usually associated with immunosuppression.
- Rare cases of hepatosplenic T-cell lymphoma (HSTCL) have occurred in patients treated with Adalimumab. This rare type of T-cell lymphoma has a very aggressive disease course and is usually fatal. Some of these HSTCL with Adalimumab have occurred in young adult patients on concomitant treatment with azathioprine or 6-mercaptopurine.
Haematologic reactions
- The following haematologic reactions have been reported with TNF-antagonists: pancytopaenia, aplastic anaemia, cytopoenia (e.g. thrombocytopaenia, leucopaenia) and blood dyscrasias (e.g. persistent fever, bruising, bleeding, pallor). Medical attention should be sought upon observation of these symptoms and Adalimumab should be discontinued.
Vaccinations
- Similar antibody responses to the standard 23-valent pneumococcal vaccine and the influenza trivalent virus vaccination were observed in a study in adult subjects with RA who were treated with Adalimumab or placebo. Therefore, patients can receive vaccination (except live vaccines) while on Adalimumab treatment.
Congestive heart failure
- Increased mortality and worsening congestive heart failure have been reported in patients treated with TNF-antagonists including Adalimumab. Treatment must be discontinued inpatients who develop new or worsening symptoms of congestive heart failure.
Autoimmune processes
- Autoantibodies may develop in patients taking Adalimumab with rare instances of symptoms of lupus-like syndrome. Adalimumab treatment should be discontinued in such cases.
Concurrent administration of TNF-antagonists or other biological DMARD
- Serious infections have been seen with concurrent use of Anakinra with Etanercept. Therefore Adalimumab is not recommended to be used with Anakinra. Adalimumab should also not be used with Infliximab, Etanercept, Abatacept, Certolizumabpegol, or Golimumab.
Small bowel obstruction
- Failure to respond to treatment for Crohn’s disease may indicate the presence of fixed fibrotic stricture that may require surgical treatment. Available data suggest that Adalimumab does not worsen or cause strictures.
Surgery
- There is little or no safety experience in patients undergoing surgical procedures or arthroplasty during Adalimumab treatment. The long half-life of Adalimumab should be considered prior to planning a surgery and the patient should be monitored for infections.
Elderly Population
- Risk of infection in elderly patients over 65 years of age is higher than those below this age with some having fatal outcomes. Thus, particular attention should be given.
INTERACTION WITH OTHER MEDICINAL PRODUCTS AND OTHER FORMS OF INTERACTION
Adalimumab treatment as monotherapy and with concomitant methotrexate has been studied in rheumatoid arthritis, polyarticular juvenile idiopathic arthritis and psoriatic arthritis patients. Adalimumab when given with methotrexate results in lower antibody formation, decreased clearance and increased efficacy of Adalimumab in comparison to Adalimumab monotherapy treatment. Combinations of Adalimumab with Anakinra, and Adalimumab with Abatacept are not recommended.
PREGNANCY AND LACTATION
Pregnancy
- Limited clinical data is available on exposed pregnancies for Adalimumab.
- Preclinical studies conducted in monkeys revealed no indication of maternal toxicity, embryotoxicity or teratogenicity; postnatal toxicity and fertility effects of Adalimumab are unavailable.
- Adalimumab can affect normal immune responses of the newborn if administered during pregnancy due to its inhibition of TNF and hence it is not recommended in pregnancy. Women should use adequate contraception during Adalimumab treatment and for a further five months after the last dose to prevent pregnancy as it bears the potential to cross the placenta into the serum of infants. This could lead to increased risk of infection to the infant. Live vaccinations to infants exposed to Adalimumab in utero is not recommended for at least 5 months after the mother’s last injection.
Lactation
- It is not recommended for women to breast-feed for at least five months after the last Adalimumab injection as human immunoglobulins are excreted in milk and there is no data suggesting whether Adalimumab is excreted in human milk or absorbed systemically after ingestion.
EFFECTS ON ABILITY TO DRIVE AND USE MACHINES
Administration of Adalimumab may result in vertigo and visual impairment which can influence the ability to drive and use machines.
ADVERSE EVENTS OBSERVED IN CHL’s CLINICAL TRIAL WERE:
Dyspnoea, Fungal infection, Gastritis, Headache, Injection site reaction, Joint swelling, Oligomenorrhoea, Pollakiuria, Polymenorrhoea, Pulmonary tuberculosis, Pyrexia, Rash, Urinary tract infection, Vomiting, Abdominal discomfort, Abdominal pain, Accelerated hypertension, Arthralgia, Asthenia, Body tinea, Chest pain, Cough, Diarrhoea and Dyspepsia.
UNDESIRABLE EFFECTS
The most commonly reported undesirable effects in patients taking Adalimumab include: infections (such as nasopharyngitis, upper respiratory tract infection and sinusitis), injection site reactions (erythema, itching, haemorrhage, pain or swelling), headache and musculoskeletal pain. Some fatal infections (including sepsis, opportunistic infections, TB), HBV reactivation and various maligancies (including leukaemia, lymphoma and HSTCL) have been reported. Serious reactions such as haematological, neurological and autoimmune reactions were also reported. Some rare reactions include pancytopenia, aplastic anaemia, central and peripheral demyelinating events, lupus, lupus-related conditions and Stevens-Johnson syndrome.
Disease Calculator
Mayo Clinic Score of Ulcerative Colitis
Truelove and Witts' severity
Truelove and Witts' severity index
Severity of ulcerative colitis
Mild, moderate and severe
In this guideline, the categories of mild, moderate and severe are used to describe ulcerative colitis:
- In adults these categories are based on the Truelove and Witts' severity index (see table 1). This table is adapted from the Truelove and Witts' criteria
- In children and young people these categories are based on the Paediatric Ulcerative Colitis Activity Index (PUCAI) (see table 2).
Table 1 Truelove and Witts' severity index
|
Mild |
Moderate |
Severe |
| Bowel movements (no. per day) |
Fewer than 4 |
4–6 |
6 or more plus at least one of the features of systemic upset (marked with * below) |
| Blood in stools |
No more than small amounts of blood |
Between mild and severe |
Visible blood |
| Pyrexia (temperature greater than 37.8°C) * |
No |
No |
Yes |
| Pulse rate greater than 90 bpm * |
No |
No |
Yes |
| Anaemia * |
No |
No |
Yes |
| Erythrocyte sedimentation rate (mm/hour) * |
30 or below |
30 or below |
Above 30 |
Table 2 Paediatric Ulcerative Colitis Activity Index (PUCAI)
Disease severity is defined by the following scores:
severe: 65 or above
moderate: 35–64
mild: 10–34
remission (disease not active): below 10
|
Item |
Points |
1. |
Abdominal pain |
|
No pain
Pain can be ignored
Pain cannot be ignored |
0
5
10 |
2. |
Rectal bleeding |
|
None
Small amount only, in less than 50% of stools
Small amount with most stools
Large amount (50% of the stool content) |
0
10
20
30 |
3. |
Stool consistency of most stools |
|
Formed
Partially formed
Completely unformed |
0
5
10 |
4. |
Number of stools per 24 hours |
|
0–2
3–5
6–8
>8 |
0
5
10
15 |
5. |
Nocturnal stools (any episode causing wakening) |
|
No
Yes |
0
10 |
6. |
Activity level |
|
No limitation of activity
Occasional limitation of activity
Severe restricted activity |
0
5
10 |
|
Sum of PUCAI (0–85) |
|
References:
- NICE (June 2013). Ulcerative colitis Management in adults, children and young people